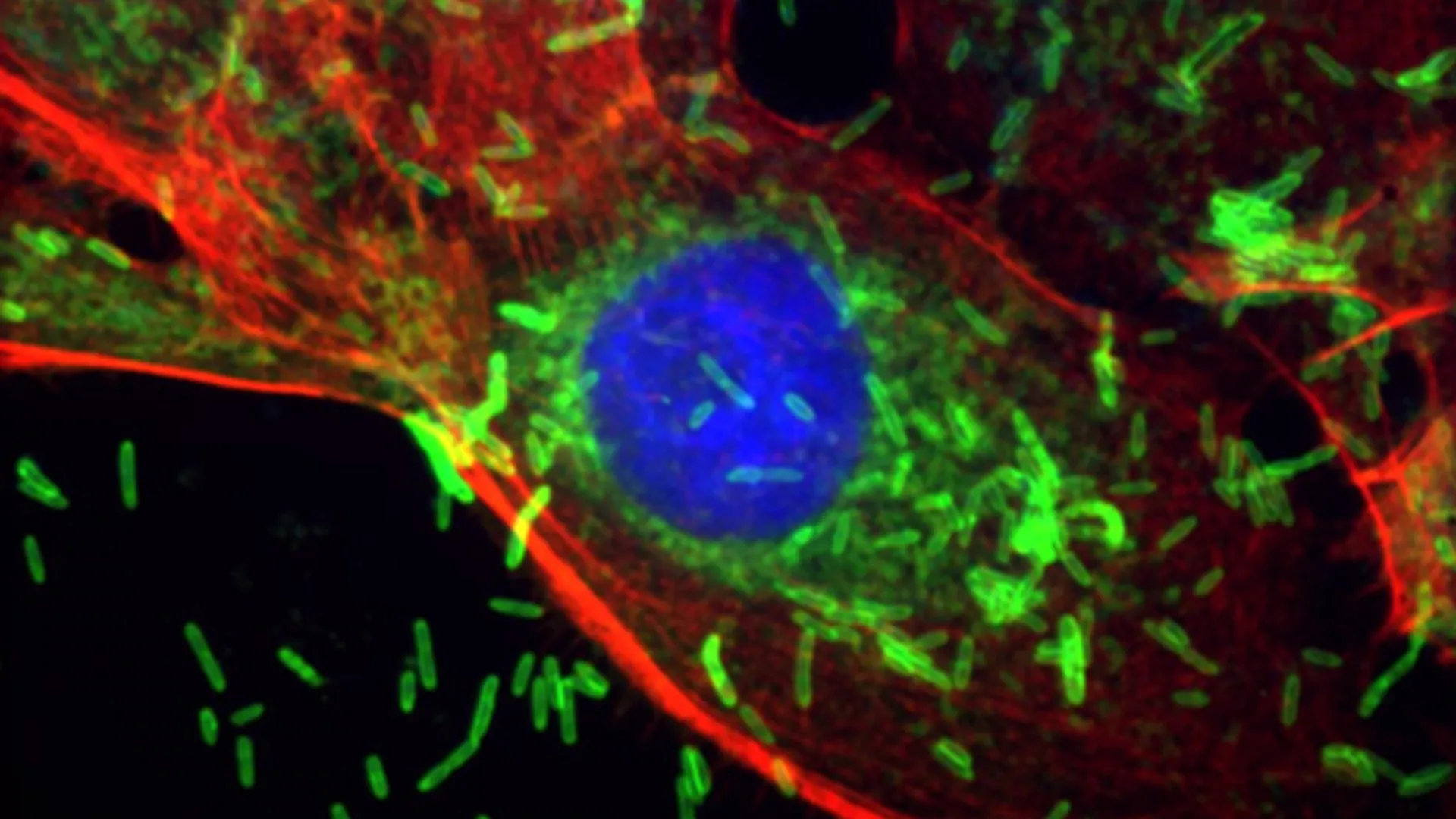
Scientists at the Johns Hopkins Kimmel Cancer Center and the Bloomberg~Kimmel Institute for Cancer Immunotherapy have identified a surprising link between oral health and breast cancer. Their research shows that a bacterium commonly associated with gum disease may help initiate breast cancer, accelerate tumor growth, and promote its spread by damaging DNA and changing how cancer cells behave.
Published in Cell Communication and Signaling, the study focuses on Fusobacterium nucleatum, a microbe previously connected to colorectal and other cancers. The researchers found that this bacterium can enter the bloodstream and settle in breast tissue, where it triggers inflammation and early cellular changes linked to cancer. In animal models of human breast cancer, the presence of this bacterium sped up tumor growth and increased the spread of cancer cells from the breast to the lungs.
“The key takeaway is that this oral microbe can reside in breast tissue and that there is a connection between this pathogen and breast cancer,” says Sharma, adding that the team’s study was inspired by many small studies that looked at thousands of patients and connected periodontal disease to breast cancer.
“We wanted to dig deeper and see if we could uncover the underlying connections,” says Sheetal Parida, Ph.D., first author and a research associate working with Sharma.
DNA Damage and Tumor-Promoting Changes
Experiments using mouse models and human breast cancer cells revealed how the bacterium affects tissue. When F. nucleatum was introduced directly into breast ducts, it caused metaplastic and hyperplastic lesions, noncancerous changes where cells either grow excessively or shift into a different type. These changes were accompanied by inflammation, DNA damage, and increased cell proliferation. When the bacterium entered the bloodstream, it significantly boosted the growth and spread of existing tumors.
The team also uncovered a key biological process behind these effects. Exposure to F. nucleatum damaged cellular DNA and activated repair systems that are prone to errors. One of these, nonhomologous end joining, rapidly reconnects broken DNA strands but can introduce mutations. Even short exposure to the bacterium increased levels of a protein called PKcs, which was linked to greater cancer cell movement, invasion, stem-like traits, and resistance to chemotherapy.
Higher Risk in BRCA1-Mutant Cells
Certain cells appeared especially susceptible. Epithelial cells (the cells that line the breast ducts) and breast cancer cells carrying BRCA1 mutations were more easily affected. These BRCA1-mutant cells had elevated levels of a surface sugar (Gal-GalNAc), which helps bacteria attach to and enter cells. As a result, these cells absorbed more F. nucleatum and retained it over time, even across multiple cell generations, intensifying DNA damage and cancer-promoting effects.
“Our findings reveal a link between oral microbes and breast cancer risk and progression, particularly in genetically susceptible individuals,” says Sharma. “Nothing happens in isolation. The results suggest that multiple risk factors come together with F. nucleatum acting as an environmental factor that may cooperate with inherited BRCA1 mutations to promote breast cancer and tumor aggressiveness.”
Implications for Oral Health and Cancer Risk
The researchers emphasize that more work is needed to understand how these findings translate to patient care. Future studies will explore whether maintaining good oral health could play a role in reducing breast cancer risk.
In addition to Sharma, the research team included Sheetal Parida, Deeptashree Nandi, Deepak Verma, Mingyang Yi, Ashutosh Yendi, Jessica Queen, Kathleen Gabrielson and Cynthia Sears.
The study was supported by the Breast Cancer Research Foundation, Congressionally Directed Medical Research Programs Department of Defense Breast Cancer Research Program grants BC191572 and BC210668), the John Fetting Fund for Breast Cancer Prevention and the Bloomberg~Kimmel Institute for Cancer Immunotherapy.